
Photo by Alex Wild
Ant species diversity and community dynamics
Ants have become an important taxonomic group for biological inventories, as they are diverse, ecologically important, easily collected, and because ant diversity is correlated with diversity of other organisms (Agosti et al. 2000). Dr. Solomon has been involved in ant inventories in Argentina, Brazil, Fiji, Panama, Peru, Cocos Island (Costa Rica), and the Big Thicket National Preserve (USA).
The Big Thicket National Preserve.

A series of invasions by non-native ants has had major impacts on the arthropod fauna of the southeastern United States (McGown et al. 2013). In particular, the red imported fire ant (Solenopsis invicta) is well known for its ability to dominate landscapes and displace native ants and other arthropods. In the last decade, the tawny crazy ant (Nylanderia fulva) has been spreading through the southeast United States and has been shown to displace S. invicta in some ecosystems (LeBrun et al. 2013, Horn et al., 2013). N. fulva is also negatively associated with arthropod richness and abundance (LeBrun et al. 2013).
A current project in collaboration with Dr. Tom E. X. Miller and undergraduate students involves: (1) characterizing the ant communities of the various units of the Big Thicket National Preserve, (2) estimating the relative proportion of native ants in each unit, (3) documenting the spread of N. fulva as it invades the Big Thicket region, (4) determining the impact of N. fulva presence on the ant community and the arthropod community in general, and (5) determining whether the size and shape of the units influences the ability of N. fulva to invade. A new avenue of research is investigating whether a citizen science approach can be used to track N. fulva outside the Big Thicket National Preserve.
A RAPID award from the National Science Foundation in 2018 has allowed Tom Miller, Sarah Bengston, and Scott Solomon to study how extreme flooding associated with Hurricane Harvey in August 2017 has affected the abundance and community composition of Big Thicket ants.
Cocos Island, Costa Rica.
Cocos Island is a small, volcanic island 500 kilometers off the coast of Costa Rica, and is the only island in the tropical eastern Pacific Ocean that supports a rainforest environment. Its isolation has given rise to several endemic species of plants, insects, birds, and lizards.
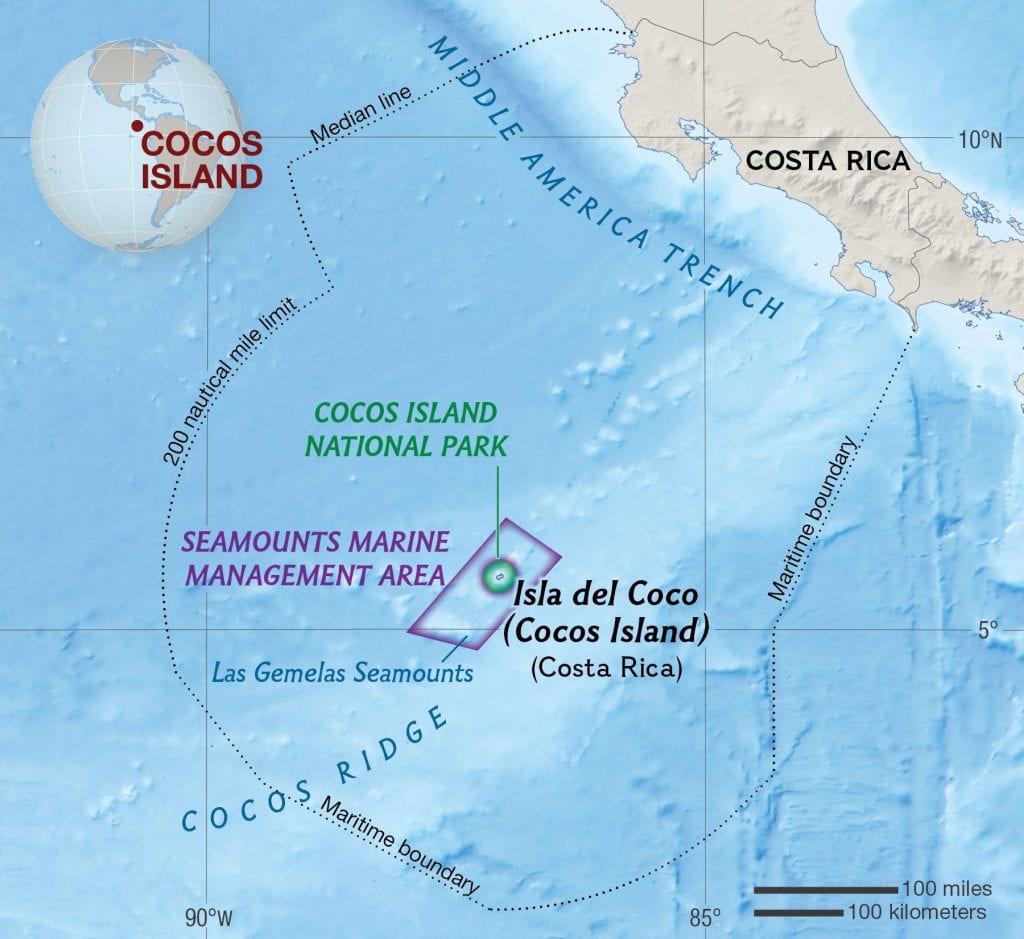
Cocos Island is located 500 km off the Pacific coast of Costa Rica (map courtesy of National Geographic)
Dr. Solomon and Dr. Alexander (Sasha) Mikheyev of the Okinawa Institute of Science and Technology and Australian National University have studied the ant fauna of Cocos Island in order to understand the balance between native and introduced species of ants.
Their systematic survey of the ants of Cocos Island, conducted in July 2003, identified 19 species of ants. Of these, the majority were widespread “tramp” species. However, one endemic species, Camponotus cocosensis, was found in abundance. Another endemic, Camponotus biolleyi, first collected by expeditions in the early 1900s, was not found; however, they collected several specimens later described by Jack Longino as a new species: Adelomyrmex coco (Longino 2012).

Adelomyrmex coco, a new species of ant endemic to Cocos Island (Photo by Jack Longino)
Systematics of the “Higher Attines”
The fungus-growing ants cultivate fungi as their primary food source. The best known are the leafcutter ants (Acromyrmex and Atta), which are considered the dominant herbivores of Central and South America. The fungi are maintained in massive underground nests that can house upwards of five million individual ants.

A mature nest of leafcutter ants (Atta laevigata) in a pine plantation in Venezuela (Photo by Bob Horth)
To understand the evolutionary history of these ants and how they have co-evolved with their symbiotic fungi, Dr. Solomon worked with Dr. Mauricio Bacci and his lab at São Paulo State University to create the first phylogenetic reconstruction of the genus Atta using nuclear and mitochondrial genes (Bacci et al. 2009).
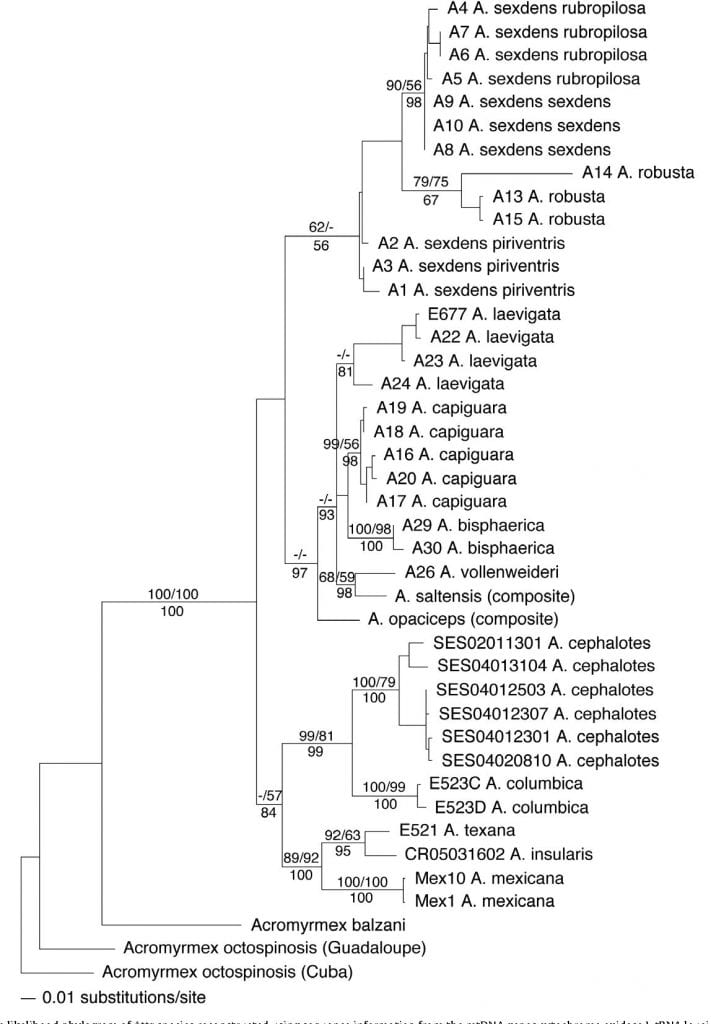
Phylogeny of Atta leafcutter ants based on mtDNA and nDNA
Leafcutter ants and their symbionts are also a fantastic model system for examining how the interactions between organisms influence the geographic range of each species. Together with Ulrich Mueller and others, Dr. Solomon showed that the Texas leafcutter ant, Atta texana, has adapted to the relatively harsh winters in northern Texas by adjusting where it houses its fungal gardens (deeper during winter) and via the evolution of cold-resistant strains of fungi. They showed that even colder winters currently prevent these ants and their symbionts from spreading further north (Mueller et al. 2011).
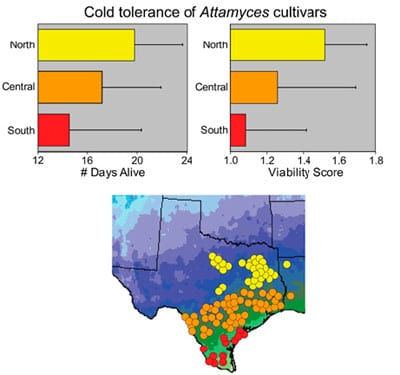
Fungi cultivated by leafcutter ants (Atta texana) at the northern edge of their range are more tolerant of cold
Unlike the leafcutters, the majority of fungus-growing ants do not typically cut leaves. Instead they use small bits of organic matter to grow their fungus gardens and have small, simple nests with only several hundred or several thousand worker ants, all of which look nearly identical. The evolutionary transition between the “lower” fungus–growing ants and the highly derived leafcutter ants is one of the most important and fascinating events in the history of the insects. By using fresh vegetation as a substrate for fungal cultivation, the ancestor of the leafcutters gained access to an incredibly abundant and reliable food source, especially in the tropical forests of South America.
Extensive sampling of the fungi cultivated by diverse leafcutter ant species throughout their geographic ranges revealed that the leafcutters do not exclusively cultivate a single clade of fungi (Mueller et al. 2017, Mueller et al. 2018). This suggests that the key to leafcutter agriculture is not simply cultivating a superior fungal cultivar.
Did the switch to fresh vegetation provided the opportunity for the ants’ colonies to grow larger and become more specialized? Or did an increase in colony size and complexity evolved first, requiring the use of an abundant and reliable food source like leaves in order to be maintained? The order of evolutionary events is key to understanding the evolutionary history of the higher attines. In order to piece together the evolutionary history of the higher attine symbiosis, Dr. Solomon has working with colleagues at the Smithsonian Institution and several universities in the USA and Brazil to complete a detailed examination of the closest living relatives to the leafcutters.
This critical position is occupied by Trachymyrmex, one of the most diverse genera of fungus-growing ants, with 49 currently recognized species. Trachymyrmex species share several biological and ecological traits with the lower attines, and others with the more derived leafcutters, emphasizing their position as transitional species. A manuscript elucidating the evolutionary relationships among this important group of attine ants and their microbial symbionts using molecular systematic techniques is currently in press at Systematic Entomology.

A Trachymyrmex ant with its fungal cultivar
In addition to the fungus kept as food for larvae and worker ants, other microbes inhabit the nests of attine ants, including specialized fungal parasites in the genus Escovopsis. Recently, thanks to our extensive sampling in Brazil, Dr. Solomon and colleagues discovered that the diversity of Escovopsis parasites found in higher attine nests is much greater than was previously thought. They documented several previously unknown lineages of Escovopsis, one of which was described as a new species (Meirelles et al. 2015). Furthermore, they found that despite high-level congruence between the evolutionary history of the ants, their mutualistic fungi, and the parasite, higher attine nests of different genera appear to share similar strains of Escovopsis, contradicting the prevailing model of co-cladogenesis (Meirelles et al. 2015).
Indeed, the nests of fungus growing ants are susceptible to growth by a wide range of microbial species, and the ants benefit from antibiotic compounds produced by actinomycete bacteria that grow on their exoskeletons. It has been suggested that such bacteria have co-evolved together with the ants, their fungal cultivars, and the parasite Escovopsis. However, Dr. Solomon and his colleagues found that bacteria in the genus Pseudonocardia in association with Trachymyrmex nests have broad inhibition activity, suggesting they are not highly specialized symbionts (Meirelles et al. 2014). Indeed, one compound they isolated from a nest of Trachymyrmex ants is effective against the fungus Candida (Mendes et al. 2013). In addition to suggesting the generalized nature of the activity of the bacteria found in attine nests, these results suggest that the compounds found in attine nests may be of medical importance.
Diversity and phylogeography of Amazonian ants
The Amazon Basin contains the greatest diversity of land-dwelling species on earth, yet there are many species that remain undiscovered. Ants are especially common in these forests. In fact, there are so many ants in the Amazon rainforest that their collective biomass exceeds that of all vertebrates combined!
Dr. Solomon’s work with Dr. Ted Schultz of the Smithsonian Institution and Dr. Heraldo Vasconcelos of the Federal University of Uberlandia in Brazil, as well as other Brazilian and American collaborators seeks to further explore the diversity of ant species in the Amazon and adjacent regions. The evolutionary reasons why this region developed such a rich biota are likewise not well understood. Several hypotheses have been proposed, each attempting to explain the origins of this hyperdiversity by proposing a different mechanism for how geographic barriers arise in what appears, at least superficially, to be a relatively homogeneous area.
Dr. Solomon and colleagues used three species of leafcutter ants (Atta cephalotes, Atta sexdens, and Atta laevigata) to test these hypotheses, employing a combination of methods from population genetics, phylogenetics, and paleodistribution modeling. The results suggested that rivers, commonly suggested as a potential barrier for terrestrial organisms, do not play a role in diversification of Amazonian leafcutter ants. They could not rule out habitat refugia during the Pleistocene or marine incursions into the Amazonian lowlands, two of the other leading hypotheses (Solomon et al. 2008).
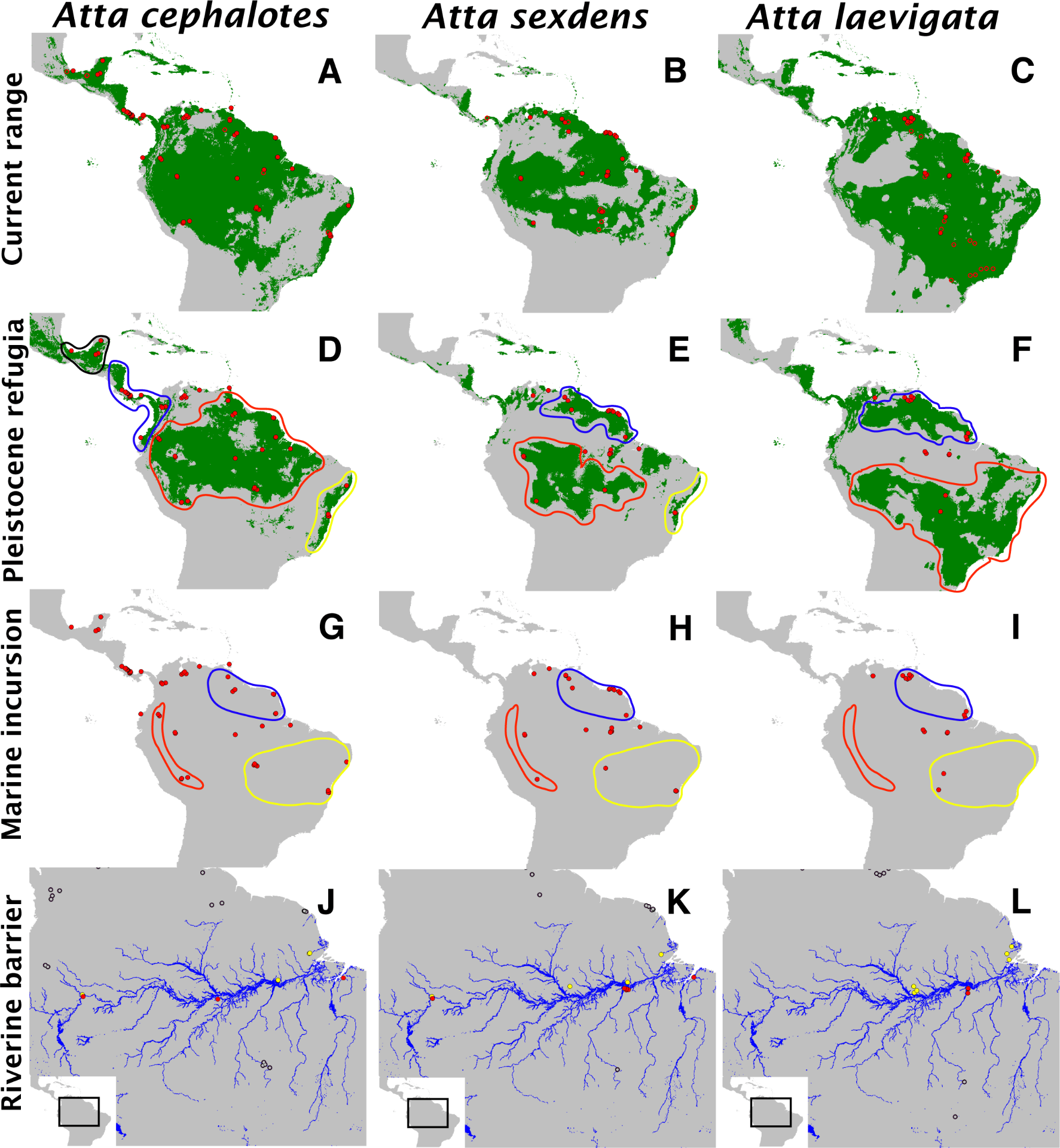
A comparison of paleodistibutions and contemporary population genetic diversity can be used to test competing hypotheses on the origins of biological diversity in the Amazon Basin
To determine whether these conclusions apply more generally, former Rice undergraduate student George Romar worked with Dr. Solomon to compile published molecular phylogenetic studies of Amazonian species. To determine whether recent speciation events have taken place entirely within Amazonia (as assumed by the hypotheses described above) or whether areas adjacent to the Amazon Basin have played a role in such speciation events, they compared the number of sister species pairs in which both species are exclusively Amazonian and those for which the sister species occurs outside of Amazonia.
Their unpublished results suggest that speciation has occurred outside of Amazonia or at its periphery nearly as often as it has occurred within Amazonia. This suggests that the prevailing hypotheses on how Amazonian diversity originated are incomplete and that neighboring regions, such as the Andes, have been important for recent speciation.